Overview
This article explains the practical difference between free chlorine and total chlorine, and how this affects sensor selection and reference testing.
Definitions
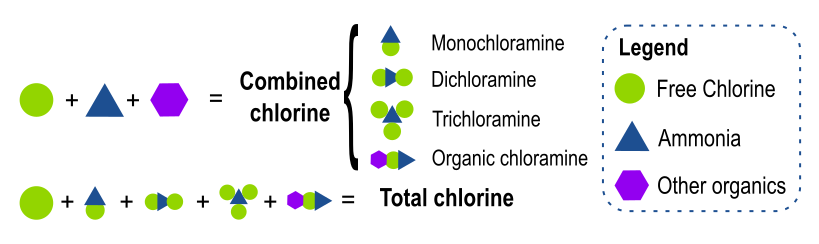
- Free chlorine: chlorine available for immediate disinfection (mainly HOCl and OCl-, and in some contexts dissolved Cl2).
- Combined chlorine: chlorine bound to nitrogen compounds, mainly chloramines (NH2Cl, NHCl2, NCl3) and organic chloramines.
- Total chlorine: sum of free chlorine and combined chlorine.
How They Are Related
When free chlorine reacts with ammonia or organic nitrogen compounds, combined chlorine forms.
Therefore: Total chlorine = Free chlorine + Combined chlorine
What Changes in Measurement
- Zirkon® DIS / ZIRKON® ONE DIS: for free chlorine measurement.
- Zirkon® DIS Total / ZIRKON® ONE TCL BIOCIDES: for total chlorine measurement.
The key difference is not only the displayed parameter, but also the sensor chemistry and matching calibration/reference method.
Reference Testing and Calibration
- Always use a reference method configured for the same parameter you measure (free or total chlorine).
- Use the correct reagents for the selected parameter.
- A mismatch between sensor parameter and DPD method leads to incorrect calibration and unstable operation.
Application Notes
- Free chlorine control is typically preferred where immediate disinfecting activity is the main focus.
- Total chlorine monitoring is typically preferred where chloramines/combined chlorine are relevant to the process.
- As a rule of thumb, free chlorine measurement is commonly applied below pH 8.0, while total chlorine applications can extend to higher pH ranges (up to approx. pH 10, depending on conditions).
Comments
0 comments
Please sign in to leave a comment.